New liver damage mechanism discovered in ischemia-reperfusion injury
A new study in Nature Communications has uncovered a molecular pathway that worsens liver damage during ischemia-reperfusion injury (IRI). The research highlights how a protein called Btg2 intensifies cell death in the liver, offering potential targets for future treatments.
Scientists found that Btg2 disrupts a protective process in liver cells, leading to increased injury after blood flow is restored. This discovery could improve outcomes for patients undergoing liver transplants or surgeries.
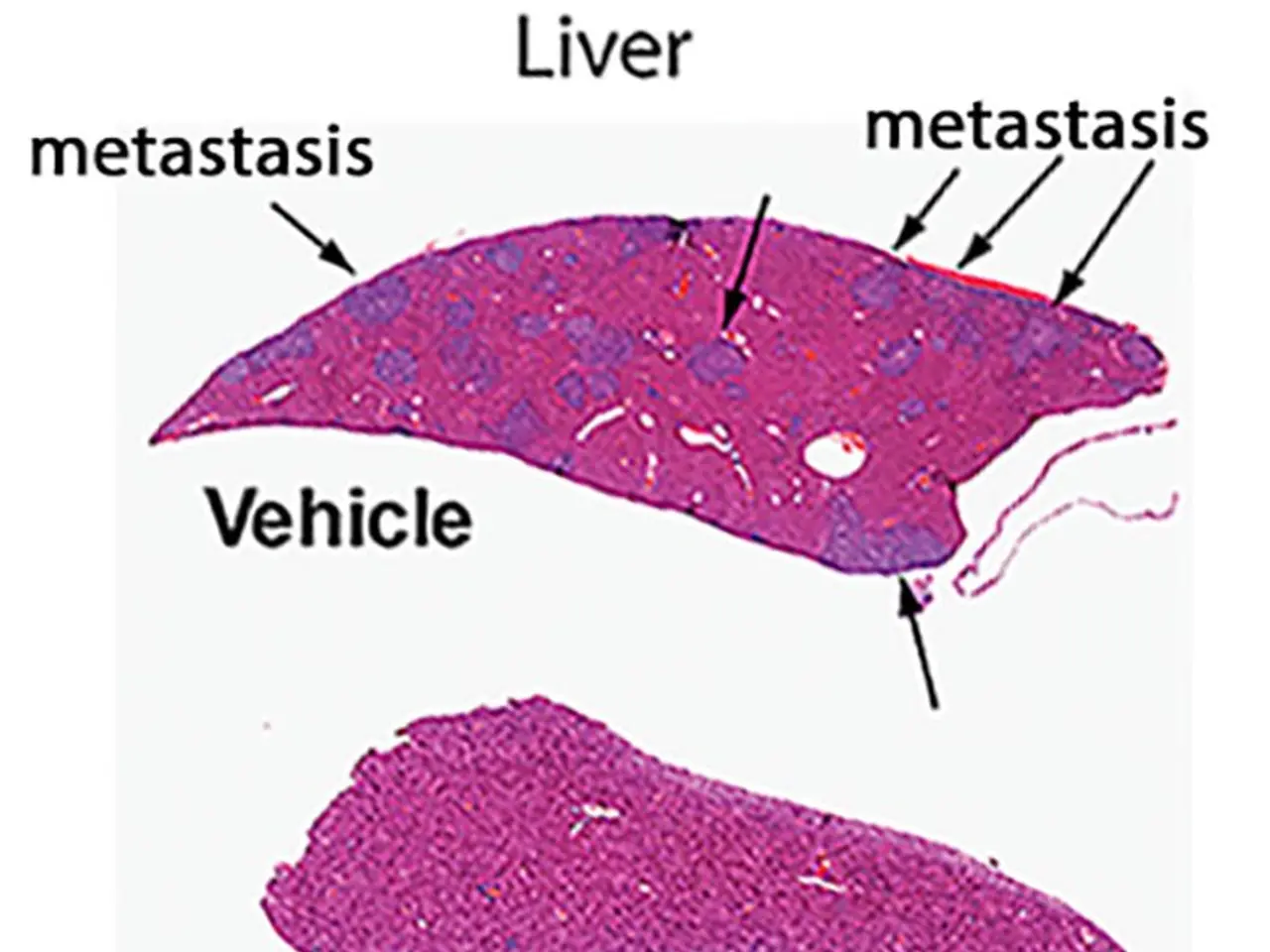
The study used mouse models to show that removing the Btg2 gene reduced liver damage after ischemia-reperfusion. Without Btg2, markers of ferroptosis (a type of iron-dependent cell death) and apoptosis (programmed cell death) were significantly lower.
Btg2 works by blocking a modification called UFMylation on another protein, Fmo1. Normally, UFMylation helps protect liver cells from oxidative damage. But when Btg2 interferes, Fmo1 loses this protection, making cells more vulnerable to irreversible injury. Further experiments revealed that increasing Btg2 levels worsened cell damage after IRI. Conversely, reducing Btg2 or boosting Fmo1 UFMylation shielded liver cells from harm. This suggests a delicate balance between survival and death in stressed hepatocytes, controlled by Btg2. The findings link oxidative stress regulation with protein modification systems, opening new directions in cell death research. The Btg2-Fmo1 interaction may also serve as a biomarker to guide personalised care for patients at risk of liver injury.
The study identifies Btg2 as a key regulator of liver damage during ischemia-reperfusion. Targeting this protein or enhancing Fmo1 UFMylation could protect liver cells in clinical settings like transplants and surgeries.
These insights may lead to better patient stratification and perioperative treatments, reducing complications from hepatic IRI.